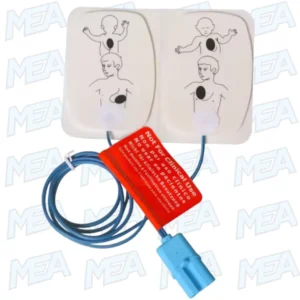
The Philips M3716A Electrode Pads are genuine, original adult/child multifunction defibrillator electrode pads designed to deliver reliable, consistent, and clinically accurate performance in life-saving situations.
Engineered by Philips to meet the highest medical standards, these electrode pads are compatible with a wide range of Philips defibrillators, including HeartStart XL+, XL, MRx, FR2, and FR3 models.
Designed for patients weighing ≥10 kg, these Philips AED electrode pads support both adult and pediatric defibrillation, making them a versatile and cost-effective solution for hospitals, emergency responders, clinics, ambulances, and critical care environments. Their radiolucent multifunction design ensures uninterrupted imaging, monitoring, pacing, and defibrillation without pad removal.
When seconds matter, using original Philips M3716A electrode pads ensures optimal energy delivery, accurate signal transmission, and dependable adhesion factors that can directly impact patient outcomes.
Why Choose Original Philips M3716A Electrode Pads?
Using genuine Philips accessories is essential for maintaining device accuracy, patient safety, and regulatory compliance. The Philips M3716A multifunction electrode pads are manufactured to exact specifications, ensuring full compatibility and consistent performance with supported Philips defibrillators.
Key advantages include:
- Factory-sealed, original Philips product
- Optimized energy conduction for effective defibrillation
- Secure adhesion for reliable ECG monitoring
- Adult and child compatibility in one pad set
- Designed for emergency, hospital, and pre-hospital use
Unlike third-party alternatives, original Philips defibrillator pads reduce the risk of device errors, poor signal quality, and ineffective shock delivery.
Overview: Philips M3716A Adult/Child Electrode Pads
The Philips M3716A electrode pads are multifunction defibrillation pads, meaning they support several critical functions using a single pad set:
- Defibrillation
- External pacing
- ECG monitoring
- Cardioversion
This multifunction capability simplifies emergency workflows and reduces the need to switch accessories during critical moments.
The pads are designed with radiolucent materials, allowing X-ray imaging without pad removal, an important feature in trauma and emergency departments where rapid diagnostics are required.
Compatible Philips Defibrillator Models
The Philips M3716A electrode pads are fully compatible with the following Philips defibrillators:
- Philips HeartStart XL+
- Philips HeartStart XL
- Philips MRx
- Philips FR2
- Philips FR3
This broad compatibility makes the M3716A pads an ideal choice for organizations operating multiple Philips defibrillator models across departments or fleets.
Adult and Child Use : One Pad Set, Greater Flexibility
One of the most valuable features of the Philips M3716A adult/child electrode pads is their dual-use design. These pads are approved for patients weighing ≥10 kg, allowing healthcare providers to treat both adults and pediatric patients using a single pad set.
This reduces inventory complexity and ensures readiness in mixed-age emergency environments such as:
- Emergency rooms
- Ambulances and EMS units
- Public access AED programs
- Clinics and outpatient centers
By eliminating the need for separate adult and pediatric pads, the Philips M3716A multifunction pads improve response time and reduce the risk of using incorrect accessories during emergencies.
Superior Adhesion for Reliable Performance
The Philips M3716A electrode pads use high-quality conductive gel and medical-grade adhesive to ensure:
- Secure skin contact
- Low impedance for effective shock delivery
- Consistent ECG signal quality
- Reduced motion artifact during monitoring
Strong adhesion is critical during defibrillation and pacing, particularly in high-stress environments where patient movement, perspiration, or rapid repositioning may occur.
Radiolucent Design for Uninterrupted Imaging
Unlike standard electrode pads, the Philips M3716A radiolucent electrode pads are specifically engineered to allow X-ray imaging without removal. This feature enables clinicians to continue life-saving therapy while performing diagnostic imaging, saving valuable time in trauma and emergency cases.
Radiolucency is especially important in:
- Emergency departments
- Intensive care units (ICU)
- Trauma centers
- Critical transport settings
Optimized for Emergency and Clinical Environments
The Philips M3716A defibrillator electrode pads are designed for professional use in demanding medical environments. Their robust construction and high-quality materials ensure dependable performance in both routine clinical monitoring and high-energy defibrillation scenarios.
Ideal use settings include:
- Hospitals and cardiac units
- Ambulances and EMS services
- Fire and rescue departments
- Clinics and diagnostic centers
- Public access defibrillation programs
Whether used in a controlled hospital setting or in the field, these Philips AED electrode pads deliver consistent and predictable results.
Ease of Application in High-Stress Situations
Clear pad labeling and intuitive placement design make the Philips M3716A electrode pads easy to apply quickly and correctly. This is critical in cardiac emergencies where every second counts.
The pads are designed to support rapid deployment by:
- Trained medical professionals
- Emergency responders
- Clinical staff
Their ergonomic design minimizes confusion and reduces the likelihood of placement errors during urgent interventions.
Single-Use, Hygienic, and Factory Sealed
Each set of Philips M3716A electrode pads is supplied factory-sealed for hygiene and safety. These pads are intended for single-patient use, helping to prevent cross-contamination and ensuring optimal adhesive and conductive performance.
Using new, unopened electrode pads is essential for:
- Maintaining infection control standards
- Ensuring effective defibrillation energy transfer
- Complying with medical device regulations
Shelf Life and Storage
The Philips M3716A adult/child electrode pads are designed with a stable shelf life when stored according to manufacturer guidelines. Proper storage ensures the adhesive and conductive gel remain effective when needed.
Recommended storage conditions include:
- Cool, dry environment
- Away from direct sunlight
- Stored in original packaging until use
Always verify the expiration date before use to ensure maximum performance and patient safety.
Why Original Philips Pads Matter for Device Safety
Philips defibrillators are calibrated and tested using original Philips accessories. Using genuine Philips M3716A electrode pads ensures:
- Accurate impedance measurement
- Correct energy delivery
- Reliable device-pad communication
- Compliance with manufacturer specifications
Non-original or incompatible pads may lead to reduced performance, inaccurate readings, or device alerts.
Key Product Specifications
- Product Name: Philips M3716A Electrode Pads
- Part Number: M3716A
- Patient Type: Adult / Child
- Minimum Patient Weight: ≥10 kg
- Design: Radiolucent multifunction pads
- Functions: Defibrillation, pacing, ECG monitoring, cardioversion
- Compatibility: XL+, XL, MRx, FR2, FR3
- Brand: Philips
- Condition: New, original, factory sealed
Applications and Use Cases
The Philips M3716A multifunction electrode pads are widely used across multiple medical disciplines and emergency response scenarios, including:
- Sudden cardiac arrest (SCA) response
- Advanced cardiac life support (ACLS)
- Cardiac monitoring and pacing
- Emergency cardioversion
- Pre-hospital emergency care
Their versatility and reliability make them a trusted accessory for critical cardiac interventions.
Ideal for Bulk Buyers and Medical Facilities
Because of their broad compatibility and adult/child functionality, Philips M3716A electrode pads are an excellent choice for:
- Hospitals standardizing accessories
- EMS fleets using Philips defibrillators
- Clinics seeking reliable OEM consumables
- Distributors supplying emergency medical equipment
Standardizing on original Philips pads helps reduce training complexity and accessory mismatches.
Frequently Asked Questions
What is the part number for these Philips electrode pads?
The official Philips part number is M3716A.
Are Philips M3716A electrode pads original Philips products?
Yes, these are genuine, original Philips electrode pads, factory sealed and manufactured to Philips specifications.
Can these pads be used for both adults and children?
Yes. The Philips M3716A electrode pads are approved for adult and child patients weighing ≥10 kg.
Which defibrillators are compatible with M3716A pads?
They are compatible with Philips HeartStart XL+, XL, MRx, FR2, and FR3 defibrillators.
Are these pads radiolucent?
Yes, the pads are radiolucent, allowing X-ray imaging without removal.
Can these pads be used for pacing and ECG monitoring?
Yes. They are multifunction electrode pads supporting defibrillation, pacing, ECG monitoring, and cardioversion.
Are these electrode pads reusable?
No. Philips M3716A electrode pads are single-use only and should be replaced after each patient use.
Do these pads come sterile and sealed?
Yes. Each set is factory sealed to ensure hygiene and optimal performance.
Why should I choose original Philips pads instead of third-party alternatives?
Original Philips pads ensure device compatibility, accurate energy delivery, reliable monitoring, and manufacturer compliance.
What is the minimum patient weight for these pads?
The minimum patient weight is 10 kilograms (≥10 kg).











Reviews
There are no reviews yet.