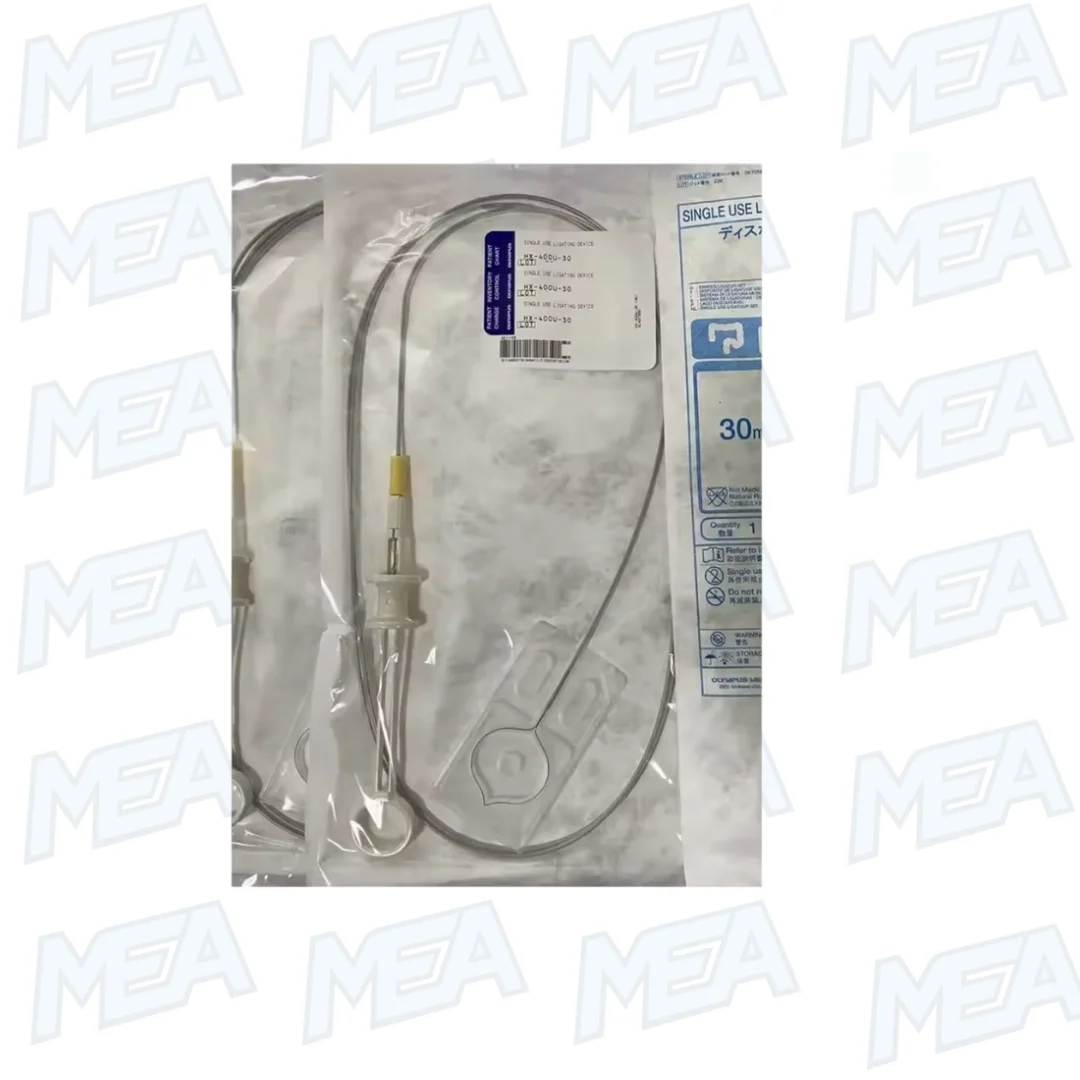
The Olympus HX-400U-30 Disposable Ligation Device is a sterile, single-use endoscopic ligating system engineered for safe and effective tissue ligation during gastrointestinal procedures.
Designed by Olympus Corporation, this medical-grade endoscope accessory supports precise polyp ligation, controlled hemostasis, and minimally invasive therapeutic interventions.
Manufactured using high-quality PVC components and supplied in sterile packaging (5 pieces per carton), the HX-400U-30 single-use ligation device is developed for consistent performance, procedural reliability, and infection-control compliance in clinical environments.
With CE quality certification and a one-year shelf life, this disposable endoscopic PolyLoop system meets the demands of modern GI units, ambulatory surgery centers, and hospital endoscopy departments.
Overview: Olympus HX-400U-30 Single-Use Ligation System
The HX-400U-30 endoscopic ligating device is engineered to provide controlled mechanical ligation of tissue structures during therapeutic endoscopy. It enables clinicians to secure pedunculated polyps or vascular lesions through a minimally invasive technique using a pre-loaded ligating loop.
Key Product Attributes
- Model Number: HX-400U-30
- Brand: Olympus
- Device Type: Disposable ligation device
- Application: Endoscopic body procedures
- Power Source: Electric (endoscopic system-assisted deployment)
- Material: Medical-grade PVC
- Quality Certification: CE certified
- Warranty: 1 Year
- Shelf Life: 1 Year
- Packaging Specification: 5 pieces per carton
- Condition: New, original
This sterile endoscopic ligature device is specifically designed to integrate seamlessly with compatible Olympus endoscope systems, ensuring precise loop deployment and secure tissue capture.
Precision Ligation for Advanced Endoscopic Procedures
The Olympus HX-400U-30 disposable ligation device is purpose-built for controlled polyp ligation and mechanical hemostasis.
It enables physicians to place a ligating loop around target tissue, tighten securely, and achieve effective vascular occlusion prior to resection or as a standalone therapeutic intervention.
Common Clinical Applications
- Ligation of pedunculated gastrointestinal polyps
- Pre-resection hemostasis control
- Endoscopic mucosal interventions
- Mechanical strangulation of targeted tissue
- Controlled ligature placement in therapeutic endoscopy
By utilizing a single-use design, the HX-400U-30 endoscopic PolyLoop reduces cross-contamination risks while ensuring consistent loop tension and deployment integrity.
Advanced Design & Material Engineering
The Olympus HX-400U-30 ligating system is manufactured from durable, medical-grade PVC and precision-engineered mechanical components. The device structure ensures smooth passage through the working channel of compatible endoscopes while maintaining optimal loop control.
Design Advantages
1. Sterile Single-Use Construction
Eliminates the need for reprocessing, reducing infection risk and improving workflow efficiency.
2. Controlled Loop Deployment
Engineered for predictable tightening and secure ligature placement.
3. Flexible Yet Durable Catheter Shaft
Supports maneuverability through the endoscope channel without compromising mechanical stability.
4. Electric Endoscopic Integration
Works in conjunction with endoscopic systems that provide mechanical control and deployment assistance.
Why Choose the Olympus HX-400U-30 Disposable Ligation Device?
When selecting an endoscopic ligating device, reliability, sterility, and brand credibility are critical. The HX-400U-30 disposable ligation system offers:
CE Quality Certification
Manufactured in compliance with European regulatory standards for medical devices.
Brand Assurance
Produced by Olympus Corporation, a globally recognized leader in endoscopy technology.
Single-Use Safety
Reduces cross-infection risk compared to reusable ligation systems.
Optimized Clinical Performance
Consistent loop deployment designed for effective mechanical hemostasis.
Convenient Packaging
Supplied as 5 sterile devices per carton for efficient inventory management.
Technical Specifications: Olympus HX-400U-30 Endoscopic PolyLoop
| Specification | Details |
| Product Name | Disposable Ligation Device |
| Model | HX-400U-30 |
| Brand | Olympus |
| Type | New, Original |
| Material | PVC |
| Application | Endoscopic body procedures |
| Power Source | Electric (system-assisted) |
| Certification | CE |
| Shelf Life | 1 Year |
| Warranty | 1 Year |
| Packaging | 5 pieces/carton |
These specifications make the Olympus HX-400U-30 single-use ligating device a dependable solution for therapeutic endoscopy procedures requiring controlled tissue ligation.
Clinical Workflow Efficiency
The disposable endoscopic ligation device streamlines procedural workflow in busy endoscopy suites. Since it is pre-sterilized and individually packaged, clinicians can:
- Open and deploy immediately without reprocessing delays
- Reduce turnaround time between procedures
- Maintain sterility standards in high-volume settings
- Improve operational efficiency
Single-use medical devices such as the HX-400U-30 are increasingly preferred in modern endoscopy practices due to enhanced infection control and predictable device integrity.
Optimized for Therapeutic Endoscopy
The Olympus HX-400U-30 ligating loop system supports minimally invasive gastrointestinal procedures. Mechanical ligation can reduce bleeding risks and assist in safer tissue resection.
Clinical Benefits
- Controlled strangulation of target tissue
- Enhanced procedural precision
- Reduced intraoperative bleeding risk
- Improved patient safety profile
- Reliable loop retention strength
Its engineered loop design ensures that once deployed, the ligature maintains consistent pressure around the targeted lesion.
Sterility & Infection Control
Infection prevention is a priority in endoscopic procedures. The Olympus HX-400U-30 disposable ligation device addresses this need through:
- Single-use design
- Sterile packaging
- CE-certified manufacturing standards
- No need for cleaning or sterilization cycles
Disposable endoscopic accessories significantly reduce contamination risk compared to reusable ligation systems.
Packaging & Storage
Each carton contains 5 sterile disposable ligation devices, individually packaged to maintain sterility until use.
Storage Recommendations
- Store in a cool, dry environment
- Avoid exposure to excessive moisture
- Do not use beyond the 1-year shelf life
- Inspect packaging integrity before use
Compatibility & System Integration
The Olympus HX-400U-30 ligation device is engineered for compatibility with appropriate Olympus endoscopic platforms. Seamless integration ensures:
- Smooth catheter insertion
- Controlled loop tightening
- Stable deployment
- Efficient procedural performance
For compatibility verification, refer to your endoscope system’s accessory guidelines.
Quality Assurance & Regulatory Compliance
The HX-400U-30 disposable endoscopic ligating device is CE certified, demonstrating compliance with essential health and safety requirements for medical devices.
Quality assurance standards include:
- Controlled manufacturing processes
- Sterility validation
- Mechanical performance testing
- Packaging integrity verification
Long-Term Value for Medical Facilities
Although designed for single use, the Olympus HX-400U-30 provides cost efficiency by:
- Reducing reprocessing costs
- Eliminating sterilization equipment burden
- Minimizing cross-contamination liability
- Improving workflow turnaround
For hospitals, clinics, and endoscopy centers, disposable ligation systems represent a strategic investment in patient safety and operational efficiency.
Ordering Information
Product Name: Olympus Disposable Ligation Device
Model: HX-400U-30
Packaging: 5 pieces per carton
Condition: New, original
Bulk ordering options may be available for institutional procurement.
Frequently Asked Questions
What is the Olympus HX-400U-30 used for?
The HX-400U-30 disposable ligation device is used for mechanical tissue ligation during gastrointestinal endoscopic procedures, including polyp management and hemostasis control.
Is the HX-400U-30 reusable?
No. This is a single-use endoscopic ligation device designed for one-time application to ensure sterility and safety.
What material is used in this ligating device?
The device is manufactured using medical-grade PVC along with precision mechanical components for secure loop deployment.
Is the Olympus HX-400U-30 CE certified?
Yes. The product carries CE certification, confirming compliance with European medical device standards.
How many devices are included per carton?
Each carton contains 5 sterile disposable ligation devices.
What is the shelf life of the device?
The shelf life is 1 year from the date of manufacture when stored properly.
Does the device require an external power source?
It operates in conjunction with compatible endoscopic systems that provide electric-assisted mechanical control.
Is the product new and original?
Yes. The HX-400U-30 supplied is new and original.
What type of procedures can it support?
It is used in therapeutic endoscopy for tissue ligation, bleeding control, and polyp management.
Why choose a disposable ligation device over a reusable one?
Disposable devices reduce cross-infection risk, eliminate sterilization requirements, and provide consistent mechanical performance.














Reviews
There are no reviews yet.